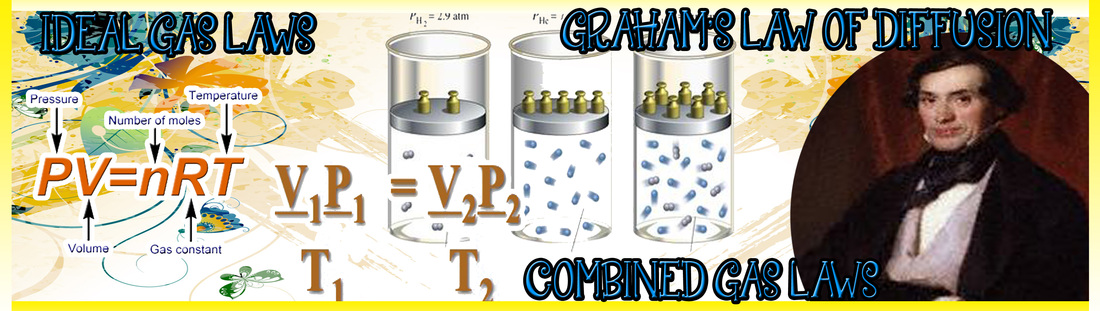
THE LAWS MADE BY ROBERT BOYLE, JACQUES CHARLES, JOSEPH GAY-LUSSAC AND AMeDEO AVOGADRO

BOYLE 's LAW
Pressure and volume are related inversely. As one variable increases, the other one decreases, vice versa. This inverse relationship between pressure and volume is known as the Boyle’s Law. Robert Boyle, a British scientist investigated and discovered this relationship.
The law states that pressure P of a sample of a gas changes inversely with the volume V as long as the temperature remains constant.
The initial volume will be multiplied to the initial pressure to give a constant value.
Initial: P1V1 = c
If the temperature would still be constant , the final pressure and final volume will be equal to a constant value.
Final: P2V2 = c
Hence, the relation of the initial and final products of the volume and the pressure of the gas may be written as:
P1V1 = P2V2
EXAMPLE:
A sample of oxygen gas has a volume of 8.0 L and a pressure of 1.0 atm. What is the new pressure if the volumeis decreased to 2.0 L?
Given:
Solution:
CHARLES 's LAW
Jacques Charles, a prominent French scientist stated the Charle’s law. He states the temperature in Kelvin and the volume of a gas are directly related when there is no change in pressure or amount of gas.
He declared that there is a direct relationship between temperature and volume of gases. It means that as one variable increases, the other one also increases. Or when one decreases, the other decreases too.
V1T2 = V2T1
REMEMBER: THE TEMPERATURE USED IN CHARLE’S LAW CALCULATION MUST BE IN KELVIN (K).
EXAMPLE:
A sample of neon gas at 760 mm Hg has a volume of 10.0 L and a temp. of 34oC. Find the new volume of the gas after the temperature has been increased to 75oC at 760 torr.
Given:
Solution:
GAY-LUSSAC 's LAW
Joseph Louis Gay-Lussac, a renowned French scientist, conducted studies on the pressure-temperature relationship of gases. He found out that the gas pressure is directly related to temperature in Kelvin. He statement eventually became known as the Gay-Lussacs law.
His law states that an increase in temperature, causes an increaseto pressure or when one variable decreases, the other one does the same as long as the volume and number of moles remain the same.
P1T2 = P2T1
One good application of this law is on sprat deodorants, etc. This law says that the pressure will increase if it is heated. This is the reason why these cans explode when heated. They were made to withstand a certain temperature and they explodes when temperature exceeds.
EXAMPLE:
10.0 L of a gas is found to exert 97.0 kPa at 25.0°C. What would be the required temperature change the pressure to standard pressure?
Given:
Note: 1.00 atm is equal to 101.325 kPa, 101 325 Pa, 760 torr and 760 mm Hg
AVOGADRO 's LAW
Amadeo Avogadro hypothesized in 1811 that the volume of a gasis directly related to the number of its moles when the temperature and pressure are held constant. It was then known as Avogadro’s law in honor for Amadeo Avogadro.
Best example of this law is a balloon. Its volume increases as air molecules are added when it’s blown. And air leaks out and its volume decreases when it is punctured.
V1N2 = V2N1
EXAMPLE:
A 6.0 L sample contains 0.5 moles of a gas. If an additional 0.25 moles of gas, what is the final total volume of the gas?
Given:
Pressure and volume are related inversely. As one variable increases, the other one decreases, vice versa. This inverse relationship between pressure and volume is known as the Boyle’s Law. Robert Boyle, a British scientist investigated and discovered this relationship.
The law states that pressure P of a sample of a gas changes inversely with the volume V as long as the temperature remains constant.
The initial volume will be multiplied to the initial pressure to give a constant value.
Initial: P1V1 = c
If the temperature would still be constant , the final pressure and final volume will be equal to a constant value.
Final: P2V2 = c
Hence, the relation of the initial and final products of the volume and the pressure of the gas may be written as:
P1V1 = P2V2
EXAMPLE:
A sample of oxygen gas has a volume of 8.0 L and a pressure of 1.0 atm. What is the new pressure if the volumeis decreased to 2.0 L?
Given:
- P1 = 1.0 atm
- V1 = 8.0 L
- P2 = ?
- V2 = 2.0 L
Solution:
- P1V1 = P2V2
- P1V1/V2 = P2
- 1.0 atm(8.0 L)/2.0L = P2
- 8.0 atm/ 2.0 = P2
- 4.0 atm = P2
CHARLES 's LAW
Jacques Charles, a prominent French scientist stated the Charle’s law. He states the temperature in Kelvin and the volume of a gas are directly related when there is no change in pressure or amount of gas.
He declared that there is a direct relationship between temperature and volume of gases. It means that as one variable increases, the other one also increases. Or when one decreases, the other decreases too.
V1T2 = V2T1
REMEMBER: THE TEMPERATURE USED IN CHARLE’S LAW CALCULATION MUST BE IN KELVIN (K).
EXAMPLE:
A sample of neon gas at 760 mm Hg has a volume of 10.0 L and a temp. of 34oC. Find the new volume of the gas after the temperature has been increased to 75oC at 760 torr.
Given:
- T1 = 34oC = 273 + 34 = 307 K
- V1 = 10.0 L
- T2 = 75oC = 273 + 75 = 348 K
- V2 = ?
Solution:
- V1T2 = V2T1
- V1T2/T1 = V2
- 10. 0 L(348 K)/307 K = V2
- 3480 L/307 = V2
- 11.3 L = V2
GAY-LUSSAC 's LAW
Joseph Louis Gay-Lussac, a renowned French scientist, conducted studies on the pressure-temperature relationship of gases. He found out that the gas pressure is directly related to temperature in Kelvin. He statement eventually became known as the Gay-Lussacs law.
His law states that an increase in temperature, causes an increaseto pressure or when one variable decreases, the other one does the same as long as the volume and number of moles remain the same.
P1T2 = P2T1
One good application of this law is on sprat deodorants, etc. This law says that the pressure will increase if it is heated. This is the reason why these cans explode when heated. They were made to withstand a certain temperature and they explodes when temperature exceeds.
EXAMPLE:
10.0 L of a gas is found to exert 97.0 kPa at 25.0°C. What would be the required temperature change the pressure to standard pressure?
Given:
- P1 = 97 kPa
- T1 = 25oC = 298 K
- P2 = 1.00 atm = 101.325 kPa
- T2 = ?
Note: 1.00 atm is equal to 101.325 kPa, 101 325 Pa, 760 torr and 760 mm Hg
- P1T2 = P2T1
- T2 = P2T1/P1
- T2 = 101.325 kPa(298 K)/7 kPa
- T2 = 311.3 K
- T2 = 310 K
AVOGADRO 's LAW
Amadeo Avogadro hypothesized in 1811 that the volume of a gasis directly related to the number of its moles when the temperature and pressure are held constant. It was then known as Avogadro’s law in honor for Amadeo Avogadro.
Best example of this law is a balloon. Its volume increases as air molecules are added when it’s blown. And air leaks out and its volume decreases when it is punctured.
V1N2 = V2N1
EXAMPLE:
A 6.0 L sample contains 0.5 moles of a gas. If an additional 0.25 moles of gas, what is the final total volume of the gas?
Given:
- V1 = 6.0 L
- N1 = 0.5 mol
- V2 = ?
- N2 = N1 + 0.25 mol = 0.75 mol
- V2 = V1N2/N1
- V2 = (6.0 L x 0.75 mol)/0.5 mol
- V2 = 4.5 L/0.5
- V2 = 9.0 L
HOW I UNDERSTAND THE IDEAL, COMBINED GAS LAWS AND GRAHAM'S LAW OF DIFFUSION

IDEAL GAS LAW
The ideal gas law is the equation of state of a hypothetical ideal gas. It is a good approximation to the behaviour of many gases under many conditions, although it has several limitations. It was first stated by Émile Clapeyron in 1834 as a combination of Boyle's law and Charles's law. The ideal gas law is often introduced in its common form:
PV= nRT
where P is the absolute pressure of the gas, V is the volume of the gas, n is the amount of substance of gas (measured in moles), T is the absolute temperature of the gas and R is the ideal, or universal, gas constant.
An ideal gas is defined as one in which all collisions between atoms or molecules are perfectly elastic and in which there are no intermolecular attractive forces. One can visualize it as a collection of perfectly hard spheres which collide but which otherwise do not interact with each other. In such a gas, all the internal energy is in the form of kinetic energy and any change in internal energy is accompanied by a change in temperature.
An ideal gas can be characterized by three state variables: absolute pressure (P), volume (V), and absolute temperature (T). The relationship between them may be deduced from kinetic theory and is called the
average kinetic energy; this invokes the idea of kinetic temperature. One mole of an ideal gas at STP occupies 22.4 liters.
COMBINED GAS LAW
The combined gas law is a gas law which combines Charles's law, Boyle's law, and Gay-Lussac's law. There is no official founder for this law; it is merely an amalgamation of the three previously discovered laws. These laws each relate one thermodynamic variable to another mathematically while holding everything else constant. Charles's law states that volume and temperature are directly proportional to each other as long as pressure is held constant. Boyle's law asserts that pressure and volume are inversely proportional to each other at fixed temperature. Finally, Gay-Lussac's law introduces a direct proportionality between temperature and pressure as long as it is at a constant volume. The inter-dependence of these variables is shown in the combined gas law, which clearly states that:
The ratio between the pressure-volume product and the temperature of a system remains constant.
This can be stated mathematically as
pV/t= K
where:
The addition of Avogadro's law to the combined gas law yields the ideal gas law.
The Combined Gas Law is a combination of Boyle's and Charles' Laws. The Combined Gas Law describes the relationship between pressure, volume, and temperature. For example, if the pressure increased, wither the volume would decrease or the temperature would increase.
The Combined Gas Law can be used to solve any Boyle's or Charles' Law problem. If one of the variables remains constant, disregard that variable by leaving it out of your equation or setting it to the same value on either side of the equals sign.
Example:
Let's try a problem with the Combined Gas Law. For example, the pressure and temperature of a gas are changed to STP (101.3 kPa/0°C) from 22.0°C and 30.8 kPa. What will be the new volume if the original volume was 205 mL?
From the problem, we know that, for the original state of the gas, the conditions were:
The unkown is V1. The equation is:
30.8 kPa x 205 mL / 295 K = 101.3 kPa x V1 / 273 K
We get:
V1 = 30.8 kPa x 205 mL / 295 K x 273 K / 101.3 kPa
V1 = 57.68 mL
However, because our problem only was to three digits of precision, the answer is:
V1 = 57.7 mL
GRAHAM'S LAW OF DIFFUSION
Graham's law, known as Graham's law of effusion, was formulated by Scottish physical chemist Thomas Graham in 1848. Graham found experimentally that the rate of effusion of a gas is inversely proportional to the square root of the mass of its particles. This formula can be written as:
(Rate1)^2(M1)=(Rate2)^2(M2)
where:
Graham's law states that the rate of effusion of a gas is inversely proportional to the square root of its molecular weight. Thus, if the molecular weight of one gas is four times that of another, it would diffuse through a porous plug or escape through a small pinhole in a vessel at half the rate of the other (heavier gases diffuse more slowly). A complete theoretical explanation of Graham's law was provided years later by the kinetic theory of gases. Graham's law provides a basis for separating isotopes by diffusion — a method that came to play a crucial role in the development of the atomic bomb.
Graham's law is most accurate for molecular effusion which involves the movement of one gas at a time through a hole. It is only approximate for diffusion of one gas in another or in air, as these processes involve the movement of more than one gas.
The ideal gas law is the equation of state of a hypothetical ideal gas. It is a good approximation to the behaviour of many gases under many conditions, although it has several limitations. It was first stated by Émile Clapeyron in 1834 as a combination of Boyle's law and Charles's law. The ideal gas law is often introduced in its common form:
PV= nRT
where P is the absolute pressure of the gas, V is the volume of the gas, n is the amount of substance of gas (measured in moles), T is the absolute temperature of the gas and R is the ideal, or universal, gas constant.
An ideal gas is defined as one in which all collisions between atoms or molecules are perfectly elastic and in which there are no intermolecular attractive forces. One can visualize it as a collection of perfectly hard spheres which collide but which otherwise do not interact with each other. In such a gas, all the internal energy is in the form of kinetic energy and any change in internal energy is accompanied by a change in temperature.
An ideal gas can be characterized by three state variables: absolute pressure (P), volume (V), and absolute temperature (T). The relationship between them may be deduced from kinetic theory and is called the
- n = number of moles
- R = universal gas constant = 8.3145 J/mol K
- N = number of molecules
- k = Boltzmann constant = 1.38066 x 10-23 J/K = 8.617385 x 10-5 eV/K
- k = R/NA
- NA = Avogadro's number = 6.0221 x 1023 /mol
average kinetic energy; this invokes the idea of kinetic temperature. One mole of an ideal gas at STP occupies 22.4 liters.
COMBINED GAS LAW
The combined gas law is a gas law which combines Charles's law, Boyle's law, and Gay-Lussac's law. There is no official founder for this law; it is merely an amalgamation of the three previously discovered laws. These laws each relate one thermodynamic variable to another mathematically while holding everything else constant. Charles's law states that volume and temperature are directly proportional to each other as long as pressure is held constant. Boyle's law asserts that pressure and volume are inversely proportional to each other at fixed temperature. Finally, Gay-Lussac's law introduces a direct proportionality between temperature and pressure as long as it is at a constant volume. The inter-dependence of these variables is shown in the combined gas law, which clearly states that:
The ratio between the pressure-volume product and the temperature of a system remains constant.
This can be stated mathematically as
pV/t= K
where:
- p is the pressure
- V is the volume
- T is the temperature measured in kelvin
- k is a constant (with units of energy divided by temperature).
- For comparing the same substance under two different sets of conditions, the law can be written as:
The addition of Avogadro's law to the combined gas law yields the ideal gas law.
The Combined Gas Law is a combination of Boyle's and Charles' Laws. The Combined Gas Law describes the relationship between pressure, volume, and temperature. For example, if the pressure increased, wither the volume would decrease or the temperature would increase.
The Combined Gas Law can be used to solve any Boyle's or Charles' Law problem. If one of the variables remains constant, disregard that variable by leaving it out of your equation or setting it to the same value on either side of the equals sign.
Example:
Let's try a problem with the Combined Gas Law. For example, the pressure and temperature of a gas are changed to STP (101.3 kPa/0°C) from 22.0°C and 30.8 kPa. What will be the new volume if the original volume was 205 mL?
From the problem, we know that, for the original state of the gas, the conditions were:
- Volume (V) = 205 mL
- Temperature (T) = 295 K
- Pressure (P) = 30.8 kPa
- Temperature1 (T1) = 273 K K
- Pressure1 (P1) = 101.3 kPa
The unkown is V1. The equation is:
30.8 kPa x 205 mL / 295 K = 101.3 kPa x V1 / 273 K
We get:
V1 = 30.8 kPa x 205 mL / 295 K x 273 K / 101.3 kPa
V1 = 57.68 mL
However, because our problem only was to three digits of precision, the answer is:
V1 = 57.7 mL
GRAHAM'S LAW OF DIFFUSION
Graham's law, known as Graham's law of effusion, was formulated by Scottish physical chemist Thomas Graham in 1848. Graham found experimentally that the rate of effusion of a gas is inversely proportional to the square root of the mass of its particles. This formula can be written as:
(Rate1)^2(M1)=(Rate2)^2(M2)
where:
- Rate1 is the rate of effusion of the first gas (volume or number of moles per unit time).
- Rate2 is the rate of effusion for the second gas.
- M1 is the molar mass of gas 1
- M2 is the molar mass of gas 2.
Graham's law states that the rate of effusion of a gas is inversely proportional to the square root of its molecular weight. Thus, if the molecular weight of one gas is four times that of another, it would diffuse through a porous plug or escape through a small pinhole in a vessel at half the rate of the other (heavier gases diffuse more slowly). A complete theoretical explanation of Graham's law was provided years later by the kinetic theory of gases. Graham's law provides a basis for separating isotopes by diffusion — a method that came to play a crucial role in the development of the atomic bomb.
Graham's law is most accurate for molecular effusion which involves the movement of one gas at a time through a hole. It is only approximate for diffusion of one gas in another or in air, as these processes involve the movement of more than one gas.
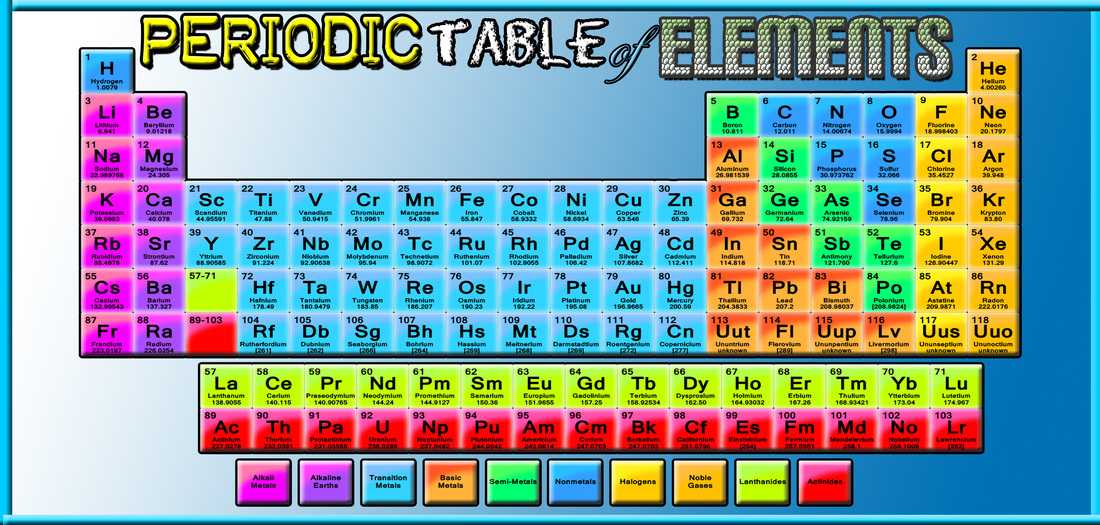
A DEEPER PERSPECTIVE ON THE PERIODIC TABLE OF ELEMENTS
The periodic table is a visual representation of the periodic law which states that certain properties of elements repeat periodically when arranged by atomic number. The table arranges elements into vertical columns (groups) and horizontal rows (periods) to display these commonalities.
The construction, arrangement and structure of the modern periodic table today reflect the growth in the understanding of chemical properties. The periodic table of the chemical elements is a tabular method of displaying the chemical elements. Although precursors to this table exist, its invention is generally credited to Russian chemist Dmitri Mendeleev in 1869. Mendeleev intended the table to illustrate recurring ("periodic") trends in the properties of the elements. The layout of the table has been refined and extended over time, as new elements have been discovered, and new theoretical models have been developed to explain chemical behavior.
The table describes the atomic structure of all elements that are known to mankind. For instance, by looking at the periodic table, a person can find out how many electrons the element has and how much it weighs. Each element has its own separate set of such data; no two elements are the same. Thus, if someone is uncertain what matter he has, he can look at the atomic structure of the material, compare it to the information in the periodic table, and identify the material by matching it to the element on the table with the same data.
How elements are arranged in the Periodic Table
The elements in the Periodic table are arranged in ascending order by proton number, which is also their atomic number.
There are eight vertical groups. Elements in each group share similar properties and have the same number of valence electrons. For example, those elements in Group 1 has one valence electron, those in group 2 has two valence electrons and so on.
The elements are also arranged horizontally in Periods. The Period of an element indicates the number of electron shells the atom has. Hydrogen and helium are in Period 1, meaning, electrons of these elements are located in only one electron shell; lithium, neon and the elements between them are in Period 2, these elements have 2 electron shells.
Metals are found on the left side of the periodic table. Non-metals are found on the right side. Elements between Group II and Group III are called transition metals. For example, iron, gold, copper, nickel and silver are all transition metals. Some elements found between metals and non-metals are metalloids, which have properties similar to both metals and non-metals.
Reading the Periodic Table
To describe the information contained within each individual box we will use a specific example: carbon.
Element Name
The purpose of the element name is obvious. However, many Periodic Tables do not include element names. For those situations you must memorize the symbols that accord to each element name.
Element Symbol
Each element has a specific one or two letter symbol that is used interchangeably with its name. These should be memorized. Most of the time, symbols quite clearly accord to the name of the element they represent, as C accords to carbon. Occasional, however, an element's name and symbol have little relation. For example, the symbol for mercury is Hg.
Atomic Number
As you move across a period the atomic number increases. Similarly, as you move down a group the atomic number increases. In this way, the atomic number represents exactly where in the periodic table an element stands.
More importantly, and the reason why the ordering of the elements according to atomic number yields elements in groups with similar chemical and physical properties, the atomic number is the same as the number of protons in the nucleus of an atom of an element, and also the same as the number of electrons surrounding the nucleus in a neutral state. Carbon, for example, has six protons and six electrons.
Atomic Mass/Weight
Along with protons, an atom also contains neutrons in its nucleus. The atomic mass (also called atomic weight) of an element is the combined number of protons and neutrons in the nucleus.
The construction, arrangement and structure of the modern periodic table today reflect the growth in the understanding of chemical properties. The periodic table of the chemical elements is a tabular method of displaying the chemical elements. Although precursors to this table exist, its invention is generally credited to Russian chemist Dmitri Mendeleev in 1869. Mendeleev intended the table to illustrate recurring ("periodic") trends in the properties of the elements. The layout of the table has been refined and extended over time, as new elements have been discovered, and new theoretical models have been developed to explain chemical behavior.
The table describes the atomic structure of all elements that are known to mankind. For instance, by looking at the periodic table, a person can find out how many electrons the element has and how much it weighs. Each element has its own separate set of such data; no two elements are the same. Thus, if someone is uncertain what matter he has, he can look at the atomic structure of the material, compare it to the information in the periodic table, and identify the material by matching it to the element on the table with the same data.
How elements are arranged in the Periodic Table
The elements in the Periodic table are arranged in ascending order by proton number, which is also their atomic number.
There are eight vertical groups. Elements in each group share similar properties and have the same number of valence electrons. For example, those elements in Group 1 has one valence electron, those in group 2 has two valence electrons and so on.
The elements are also arranged horizontally in Periods. The Period of an element indicates the number of electron shells the atom has. Hydrogen and helium are in Period 1, meaning, electrons of these elements are located in only one electron shell; lithium, neon and the elements between them are in Period 2, these elements have 2 electron shells.
Metals are found on the left side of the periodic table. Non-metals are found on the right side. Elements between Group II and Group III are called transition metals. For example, iron, gold, copper, nickel and silver are all transition metals. Some elements found between metals and non-metals are metalloids, which have properties similar to both metals and non-metals.
Reading the Periodic Table
To describe the information contained within each individual box we will use a specific example: carbon.
Element Name
The purpose of the element name is obvious. However, many Periodic Tables do not include element names. For those situations you must memorize the symbols that accord to each element name.
Element Symbol
Each element has a specific one or two letter symbol that is used interchangeably with its name. These should be memorized. Most of the time, symbols quite clearly accord to the name of the element they represent, as C accords to carbon. Occasional, however, an element's name and symbol have little relation. For example, the symbol for mercury is Hg.
Atomic Number
As you move across a period the atomic number increases. Similarly, as you move down a group the atomic number increases. In this way, the atomic number represents exactly where in the periodic table an element stands.
More importantly, and the reason why the ordering of the elements according to atomic number yields elements in groups with similar chemical and physical properties, the atomic number is the same as the number of protons in the nucleus of an atom of an element, and also the same as the number of electrons surrounding the nucleus in a neutral state. Carbon, for example, has six protons and six electrons.
Atomic Mass/Weight
Along with protons, an atom also contains neutrons in its nucleus. The atomic mass (also called atomic weight) of an element is the combined number of protons and neutrons in the nucleus.
ARE YOU A METAL, NON-METAL OR METALLOID?

Using the periodic table, you can classify the elements in many ways. One useful way is by classifying the elements through its main types: the METALS (blue in the table), NONMETALS (orange) and METALLOIDS/SEMI-METALS (green).
Most elements are metals. METALS are generally shiny, very dense, malleable, hard, and only melt at high temperatures. Metals will corrode, gradually wearing away, like rusting iron. Metals are also good conductors of electricity. Examples of metals are Gold, Silver, Gallium, Iron, Uranium, and Zinc.
The elements at the right side of the periodic table are called NONMETALS. Their surface is dull and they don’t conduct heat and electricity. As compared to metals, they have low density and will melt at low temperatures. The shape of nonmetals cannot be changed easily because they are brittle and will break. Example non-metal elements are Hydrogen, Carbon, Sulfur and Bromine.
Elements that have properties of both metals and nonmetals are called METALLOIDS/SEMIMETALS. They can be shiny or dull and their shape is easily changed. Electricity and heat can travel through metalloids but not as easily as they travel through metals. The elements recognized as metalloids are Boron, Silicon, Germanium, Arsenic, Polonium, Antimony and Tellurium.
Most elements are metals. METALS are generally shiny, very dense, malleable, hard, and only melt at high temperatures. Metals will corrode, gradually wearing away, like rusting iron. Metals are also good conductors of electricity. Examples of metals are Gold, Silver, Gallium, Iron, Uranium, and Zinc.
The elements at the right side of the periodic table are called NONMETALS. Their surface is dull and they don’t conduct heat and electricity. As compared to metals, they have low density and will melt at low temperatures. The shape of nonmetals cannot be changed easily because they are brittle and will break. Example non-metal elements are Hydrogen, Carbon, Sulfur and Bromine.
Elements that have properties of both metals and nonmetals are called METALLOIDS/SEMIMETALS. They can be shiny or dull and their shape is easily changed. Electricity and heat can travel through metalloids but not as easily as they travel through metals. The elements recognized as metalloids are Boron, Silicon, Germanium, Arsenic, Polonium, Antimony and Tellurium.
THE ATOMIC THEORIES THAT MADE AN IMPACT ON MODERN SCIENCE

An atom is the smallest particle and the basic unit of matter but ever wonder where and when was the atom discovered and how did it developed or evolved throughout the years? Well now, that question in your mind can be answered.
Truly, the atom has been quite a while but the earliest accounts of the discovery of the atom was from Democritus, a Greek philosopher. Based from his studies, he called the smallest particle of a material "Atomos", now known as the atom meaning cannot be cut. According to Democritus, all things are made of atoms, indestructible and always will be. His atomic model is composed of a circle because back then, they have no advanced technology, they just merely rely on their observations, ideas and hypothesis.
The next person who contributed to the atomic development is Aristotle known for his extreme wits and high IQ. He also made an atomic theory. His theory suggests that all things are composed mainly from 4 different elements, fire, water, earth and air. Also, he formulated another element which the stars are from, he called it "aither". Moreover, matter has for qualities mainly dryness, hotness, coldness and wetness. But despite his high intelligence, his theory was proven wrong centuries after. Maybe it is because they lack in technological advancement and they do not have sufficient materials for further studies and analysis.
Another one is Ernest Rutherford. His atomic model suggests that the atom is comprised of a single positive nucleus surrounded by negative orbiting electrons. Also, it suggests that most of the mass of the atoms are contained in the small nucleus and the rest of the atom is mostly empty space or 99.9%. This conclusions were derived from his experiment using gold foil with alpha particles particularly Helium nuclei.
Next is the famous John Dalton. His atomic theory is based on experimental observations. The postulates of his theory are stated in the following statement. "All matter is made of atoms, atoms are indivisible and indestructible, all atoms of a given element are identical in mass and properties, compounds are formed by a combination of two or more different kinds of atoms, a chemical reaction is a rearrangement of atoms and atoms can be neither created nor destroyed."
Neils Bohr is credited for the formulation of the most accurate atomic theory of all. His theory became the foundation of the modern atomic theory.His studies mainly focus on electrons. He said that electrons or the negatively charged particles orbit the nucleus without losing energy; they could move only in fixed orbits of specific energies and electrons with low energy would orbit closer to the nucleus while electrons with high energy orbit further from the nucleus.
The Eugen Goldstein came-in. His studies about the atom are derived from the experiments in his anode rays. He discovered the Canal Rays in 1886. Also the discovery which states that the cathode rays were not the only rays that will move in the opposite direction of the electrons and that the Canal Rays do the same. Canal Rays are positively charged protons and produced a reddish light at the top of the tube and a green color when the electrons hit the sides of the tube. This discovery led to the discovery of the proton.
Next is Sir James Chadwick mainly known for his discovery of the neutron in the nucleus. He disagreed with the atomic model of Etrnest Rutherford because it stated that the nucleus contains only the protons and not nothing else. He stated that with the protons are neutrons which are particles with no charge at all orbited by electrons.
And the latest atomic theory was called the Quantum model from the collaborative efforts of Werner Heisenberg, Louis de Broglie and Erwin Schrödinger. This theory mainly focused on the s shell, p shell, d shell and f shell. And also it made an introduction to Quantum numbers particularly angular, magnetic and spin quantum numbers.
These are some people who spent their time researching answers for our questions in the future. Even though that they passed-away years and years ago, they should be proud of what they have done to modern science, they made a huge impact to our lives by explaining how things are made-off. So truly from Democritus to Heisenberg et al., science and the formulation of the atomic theory has indeed come a long way!
Truly, the atom has been quite a while but the earliest accounts of the discovery of the atom was from Democritus, a Greek philosopher. Based from his studies, he called the smallest particle of a material "Atomos", now known as the atom meaning cannot be cut. According to Democritus, all things are made of atoms, indestructible and always will be. His atomic model is composed of a circle because back then, they have no advanced technology, they just merely rely on their observations, ideas and hypothesis.
The next person who contributed to the atomic development is Aristotle known for his extreme wits and high IQ. He also made an atomic theory. His theory suggests that all things are composed mainly from 4 different elements, fire, water, earth and air. Also, he formulated another element which the stars are from, he called it "aither". Moreover, matter has for qualities mainly dryness, hotness, coldness and wetness. But despite his high intelligence, his theory was proven wrong centuries after. Maybe it is because they lack in technological advancement and they do not have sufficient materials for further studies and analysis.
Another one is Ernest Rutherford. His atomic model suggests that the atom is comprised of a single positive nucleus surrounded by negative orbiting electrons. Also, it suggests that most of the mass of the atoms are contained in the small nucleus and the rest of the atom is mostly empty space or 99.9%. This conclusions were derived from his experiment using gold foil with alpha particles particularly Helium nuclei.
Next is the famous John Dalton. His atomic theory is based on experimental observations. The postulates of his theory are stated in the following statement. "All matter is made of atoms, atoms are indivisible and indestructible, all atoms of a given element are identical in mass and properties, compounds are formed by a combination of two or more different kinds of atoms, a chemical reaction is a rearrangement of atoms and atoms can be neither created nor destroyed."
Neils Bohr is credited for the formulation of the most accurate atomic theory of all. His theory became the foundation of the modern atomic theory.His studies mainly focus on electrons. He said that electrons or the negatively charged particles orbit the nucleus without losing energy; they could move only in fixed orbits of specific energies and electrons with low energy would orbit closer to the nucleus while electrons with high energy orbit further from the nucleus.
The Eugen Goldstein came-in. His studies about the atom are derived from the experiments in his anode rays. He discovered the Canal Rays in 1886. Also the discovery which states that the cathode rays were not the only rays that will move in the opposite direction of the electrons and that the Canal Rays do the same. Canal Rays are positively charged protons and produced a reddish light at the top of the tube and a green color when the electrons hit the sides of the tube. This discovery led to the discovery of the proton.
Next is Sir James Chadwick mainly known for his discovery of the neutron in the nucleus. He disagreed with the atomic model of Etrnest Rutherford because it stated that the nucleus contains only the protons and not nothing else. He stated that with the protons are neutrons which are particles with no charge at all orbited by electrons.
And the latest atomic theory was called the Quantum model from the collaborative efforts of Werner Heisenberg, Louis de Broglie and Erwin Schrödinger. This theory mainly focused on the s shell, p shell, d shell and f shell. And also it made an introduction to Quantum numbers particularly angular, magnetic and spin quantum numbers.
These are some people who spent their time researching answers for our questions in the future. Even though that they passed-away years and years ago, they should be proud of what they have done to modern science, they made a huge impact to our lives by explaining how things are made-off. So truly from Democritus to Heisenberg et al., science and the formulation of the atomic theory has indeed come a long way!
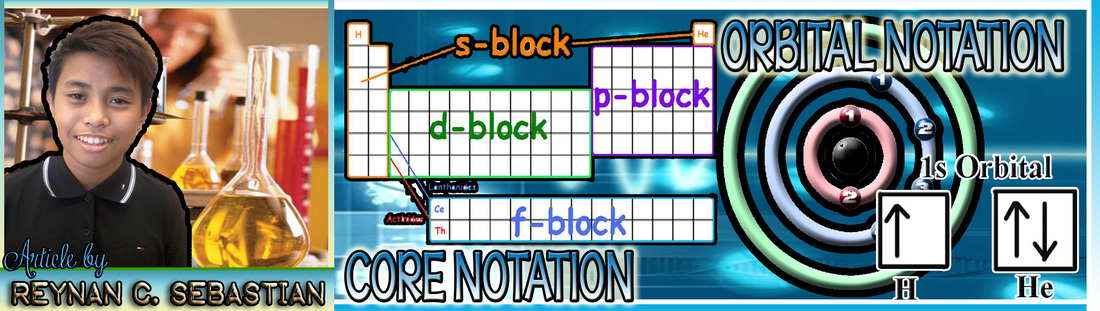
THE THINGS THEY CALL "ATOMIC NOTATION" AND "MNEMONIC DEVICE"

THE ATOMIC NOTATION
Atomic Notation, which is also known as Nuclear Notation, is a way to represent an element by showing its mass number and atomic number. The element symbol is accompanied by two smaller numbers, one at the lower left and one at the upper left part. The number on the lower portion is the atomic number; the number on the upper portion is the mass number.
It is written in Figure 1
X stands for the symbol while A represents the atomic mass and Z represents the atomic number.
The atomic mass represents the total mass or number of protons and neutrons in the nucleus. The atomic mass can be found at the upper right portion of the block/box where the element is located. Its unit is amu.
The atomic number determines the total number of protons found inside the nucleus of an atom. In the Periodic Table of Elements, the atomic number is found at the upper left portion of the block/ box where the element is located.
NOTE: A stable atom has ZERO charge because it is neutral.
For example, the element Radon has an atomic mass of 222 amu and an atomic number of 86, it is written in Figure 3.
THE MNEMONIC DEVICE
The energy levels or shells of an atom is divided into subshells namely s (Sharp), p (Principal), d (Diffuse) and f (Fundamental).
The s subshell contains a total number of two electrons or less.
The p subshell has six or less number of electrons.
The d subshell encloses ten or less electrons.
The f subshell consists of fourteen or less number of electrons.
The Mnemonic device is the object we see at the lower left part of the Periodic Table of Elements. It is an illustration on how we distribute the sublevels accordingly to the element it designates. It contains circles with the energy sublevel and arrows that indicates the next sublevel to be written during the distribution of electrons.
It takes time to memorize the sequence electron distribution, but by means of looking at the Mnemonic device, we can identify the correct sequence on how we distribute the electrons. The Mnemonic device is easy to draw and in that case we can have our own guides without the use of the Periodic Table of Elements.
For our example, we have to get the electron configuration of the element which has 22 electrons in its nucleus (Titanium), we just have to follow the arrows of the Mnemonic Device shown on Figure 2 and write the correct distribution.
1s2 2s2 2p6 3s2 3p6 4s2 3d2
Note: Sometimes, we have to cut the number of electrons of the last subshell to have the correct number of electrons.
To check, add the number of electrons and see if it is correct or incorrect.
2 + 2 + 6 + 2 + 6 + 2 + 2 = 22 e-
Atomic Notation, which is also known as Nuclear Notation, is a way to represent an element by showing its mass number and atomic number. The element symbol is accompanied by two smaller numbers, one at the lower left and one at the upper left part. The number on the lower portion is the atomic number; the number on the upper portion is the mass number.
It is written in Figure 1
X stands for the symbol while A represents the atomic mass and Z represents the atomic number.
The atomic mass represents the total mass or number of protons and neutrons in the nucleus. The atomic mass can be found at the upper right portion of the block/box where the element is located. Its unit is amu.
The atomic number determines the total number of protons found inside the nucleus of an atom. In the Periodic Table of Elements, the atomic number is found at the upper left portion of the block/ box where the element is located.
NOTE: A stable atom has ZERO charge because it is neutral.
For example, the element Radon has an atomic mass of 222 amu and an atomic number of 86, it is written in Figure 3.
THE MNEMONIC DEVICE
The energy levels or shells of an atom is divided into subshells namely s (Sharp), p (Principal), d (Diffuse) and f (Fundamental).
The s subshell contains a total number of two electrons or less.
The p subshell has six or less number of electrons.
The d subshell encloses ten or less electrons.
The f subshell consists of fourteen or less number of electrons.
The Mnemonic device is the object we see at the lower left part of the Periodic Table of Elements. It is an illustration on how we distribute the sublevels accordingly to the element it designates. It contains circles with the energy sublevel and arrows that indicates the next sublevel to be written during the distribution of electrons.
It takes time to memorize the sequence electron distribution, but by means of looking at the Mnemonic device, we can identify the correct sequence on how we distribute the electrons. The Mnemonic device is easy to draw and in that case we can have our own guides without the use of the Periodic Table of Elements.
For our example, we have to get the electron configuration of the element which has 22 electrons in its nucleus (Titanium), we just have to follow the arrows of the Mnemonic Device shown on Figure 2 and write the correct distribution.
1s2 2s2 2p6 3s2 3p6 4s2 3d2
Note: Sometimes, we have to cut the number of electrons of the last subshell to have the correct number of electrons.
To check, add the number of electrons and see if it is correct or incorrect.
2 + 2 + 6 + 2 + 6 + 2 + 2 = 22 e-
A CLOSER LOOK AT THE CORE AND ORBITAL NOTATIONS OF ELEMENTS
THE EASY YET CONVENIENT CHARACTERISTICS OF QUANTUM NUMBERS

Quantum Numbers- a set of four number that describe the values and characteristic of some property of electrons found in an orbital, their numbers are called quantum numbers.
There is also a theory named quantum mechanics says that electrons retain in quantized energy level and are arranged according to size and shape Also acc. to Werner Heisenberg it is not possible to determine the exact position and energy level of electrons
As stated there are 4 values of quantum numbers. The first one is the Principal energy level it is represented as n. The higher the value of n, the further it is from the nucleus or it simply states the energy level of an electron. For ex. 6s; 6, 3p; 3.
The allowed numbers for value of n is just 1-7.
Let's proceed to the second quantum number or the orbital angular movement represented by l. The only allowed value for l is 1-4. It determines the shape of the orbital. There are fixed numbers for each sub-shells as shown: s=0, p=1, d=2, and f=3.
The third quantum number is the magnetic quantum number, presented as ml. It is the number of orbitals and their orientation within the subshells. The assigned values are:
s=0 p=-1, 0, +1, d=-2,-1,0,+1,+2, f=-3,-2,-1,0,+1,+2,+3. The value can be determined as you draw the orbital notation. When you distribute the electrons, distribute singly pairing, mark the orbital in ascending order with the possible values.
Where the last electron drops, that will the value for ml.
And the last quantum number, or the electron spin quantum number also written as ms. It designates the direction of the electron spin of +1/2 or -1/2. When the ms is positive, the electron is refered to spin up and when it is negative, it spins down.
It is important to know the 4th quantum number because it is used to determine if the atoms have the ability to generate magnetic field. To get the value of the ms, it will be +1/2 if the last electron placed in the orbital notation was left unpaired.
And it will be -1/2 if the last electron dropped on the orbital notation was being paired with other electron on the same orbital.
And that's all about the four quantum numbers.
There is also a theory named quantum mechanics says that electrons retain in quantized energy level and are arranged according to size and shape Also acc. to Werner Heisenberg it is not possible to determine the exact position and energy level of electrons
As stated there are 4 values of quantum numbers. The first one is the Principal energy level it is represented as n. The higher the value of n, the further it is from the nucleus or it simply states the energy level of an electron. For ex. 6s; 6, 3p; 3.
The allowed numbers for value of n is just 1-7.
Let's proceed to the second quantum number or the orbital angular movement represented by l. The only allowed value for l is 1-4. It determines the shape of the orbital. There are fixed numbers for each sub-shells as shown: s=0, p=1, d=2, and f=3.
The third quantum number is the magnetic quantum number, presented as ml. It is the number of orbitals and their orientation within the subshells. The assigned values are:
s=0 p=-1, 0, +1, d=-2,-1,0,+1,+2, f=-3,-2,-1,0,+1,+2,+3. The value can be determined as you draw the orbital notation. When you distribute the electrons, distribute singly pairing, mark the orbital in ascending order with the possible values.
Where the last electron drops, that will the value for ml.
And the last quantum number, or the electron spin quantum number also written as ms. It designates the direction of the electron spin of +1/2 or -1/2. When the ms is positive, the electron is refered to spin up and when it is negative, it spins down.
It is important to know the 4th quantum number because it is used to determine if the atoms have the ability to generate magnetic field. To get the value of the ms, it will be +1/2 if the last electron placed in the orbital notation was left unpaired.
And it will be -1/2 if the last electron dropped on the orbital notation was being paired with other electron on the same orbital.
And that's all about the four quantum numbers.
references of information and pictures
- WE would like to acknowledge Google.com for all the pictures and valid resource information for the completion of the articles.
- Especially we would also like to thank www.chemistry.about.com and www.wikipedia .com.
- For the pictures of our members we would like to the cite the source which is www.facebook.com
MY OVERVIEW On CHEMISTRY

Chemistry is a branch of physical science which
deals with the study of the composition, properties, and behavior of matter.
The atom is the basic unit of Chemistry. It is concerned with atoms and their
relation to other atoms, especially on the properties of the chemical bonds
assembled between species. It also deals with the interactions betwixt
molecules and atoms and different forms of energy like photochemical reactions,
oxidation-reduction reactions, and changes in phases of matter, separation of
mixture, properties of polymers, and so on.
Chemistry is consistently called as “the central science” for it connects other natural sciences (e.g. physics, geology and biology)
There are many branches of chemistry but there are only five main branches which are organic chemistry, inorganic chemistry, analytical chemistry, physical chemistry and biochemistry.
*Organic Chemistry is concerned in the study of carbon and its compounds. It is also the study of the chemistry of life.
Organic chemistry is relevant because it is the study of life and all of the chemical reactions associated to life.
*Inorganic Chemistry deals with the study of compounds which are not covered by organic chemistry. It is the study of inorganic compounds (compounds which do not contain a Carbon- Hydrogen bond). Many inorganic compounds are those which contain metals.
*Analytical Chemistry is the study of the chemistry of matter and the evolution or development of tools used to measure properties of matter. It deals with the separation, identification, and quantification of the chemical components of natural and artificial materials
*Physical Chemistry deals with the study of macroscopic, atomic, subatomic, and particulate phenomena in chemical systems with regards to the laws and concepts of physics. It applies physics to the study of chemistry. It frequently includes the applications of thermodynamics and quantum mechanics to chemistry.
*Biochemistry is the study of chemical processes that takes place inside of living organisms. It is concerned with the chemical and photochemical processes that occur within living organisms.
Chemistry is consistently called as “the central science” for it connects other natural sciences (e.g. physics, geology and biology)
There are many branches of chemistry but there are only five main branches which are organic chemistry, inorganic chemistry, analytical chemistry, physical chemistry and biochemistry.
*Organic Chemistry is concerned in the study of carbon and its compounds. It is also the study of the chemistry of life.
Organic chemistry is relevant because it is the study of life and all of the chemical reactions associated to life.
*Inorganic Chemistry deals with the study of compounds which are not covered by organic chemistry. It is the study of inorganic compounds (compounds which do not contain a Carbon- Hydrogen bond). Many inorganic compounds are those which contain metals.
*Analytical Chemistry is the study of the chemistry of matter and the evolution or development of tools used to measure properties of matter. It deals with the separation, identification, and quantification of the chemical components of natural and artificial materials
*Physical Chemistry deals with the study of macroscopic, atomic, subatomic, and particulate phenomena in chemical systems with regards to the laws and concepts of physics. It applies physics to the study of chemistry. It frequently includes the applications of thermodynamics and quantum mechanics to chemistry.
*Biochemistry is the study of chemical processes that takes place inside of living organisms. It is concerned with the chemical and photochemical processes that occur within living organisms.
Why is chemistry so important anyway?

Chemistry is important in everyday life because almost everything is made of chemicals. Many of the changes you observe in the world around you are caused by chemical reactions. Examples include changing colors of leaves, cooking food and getting clean. Knowing chemistry can help you make a day to day decision that affects your life. Can I mix these household chemicals? What are safe mosquito repellents? Will my bottled water expire? These are some questions that chemistry can answer. Chemistry also helps our lives in agriculture, medicine, health and sanitary, energy, travel and exploration, transportation and communication, food production and clothing.
Chemistry has a big role in clothing through discovering and inventing products that help clothes to become soft, fragrant and clean against germs and also it helps in the production of clothing fabrics, some examples are dyes of different colors, fire-proof gloves, synthetic fibers, nylon threads and Waterproof Clothing. It is also important in transportation and communication through upgrading and making the communication and transportation faster and easier by upgrading the roads and developing updates with technology and application of chemistry example asphalt, tires, diesel, car engines, the modern day internet, and car batteries.
Health and sanitation made advancement throughout the years and now, some discoveries make us healthier and cleaner through the help of Chemistry by inventing and discovering such disinfectants, skin cares products, healthy water with minerals, and also antibiotics. Chemistry is important in agriculture through improving our crops, discovering and developing many kinds of fertilizer and pesticide. Also, chemistry produced more growth-enhancing feeds for our farm livestock like more yield of eggs, milk etc.
Chemistry also helps shelter by making the house or shelter more beautiful, durable and stable example paints, lacquer, termite killer, varnish, insulation. Chemistry has played a big role in energy production because it helps invent and make more efficient, more effective but cheap renewable energy resources like biomass fuel and that is good for the ecosystem. Other examples of contributions of chemistry to the field of energy production are bioethanol, solar cell, insulators and gas tank.
So therefore, chemistry plays a vital role in our day to day living, it also enhances our minds for future endeavors and lastly, it can serve as an inspiration to other people in determining what field in science to prioritize because chemistry links all the sciences. Lastly, chemistry has made countless innovations for the sake of mankind.
Chemistry has a big role in clothing through discovering and inventing products that help clothes to become soft, fragrant and clean against germs and also it helps in the production of clothing fabrics, some examples are dyes of different colors, fire-proof gloves, synthetic fibers, nylon threads and Waterproof Clothing. It is also important in transportation and communication through upgrading and making the communication and transportation faster and easier by upgrading the roads and developing updates with technology and application of chemistry example asphalt, tires, diesel, car engines, the modern day internet, and car batteries.
Health and sanitation made advancement throughout the years and now, some discoveries make us healthier and cleaner through the help of Chemistry by inventing and discovering such disinfectants, skin cares products, healthy water with minerals, and also antibiotics. Chemistry is important in agriculture through improving our crops, discovering and developing many kinds of fertilizer and pesticide. Also, chemistry produced more growth-enhancing feeds for our farm livestock like more yield of eggs, milk etc.
Chemistry also helps shelter by making the house or shelter more beautiful, durable and stable example paints, lacquer, termite killer, varnish, insulation. Chemistry has played a big role in energy production because it helps invent and make more efficient, more effective but cheap renewable energy resources like biomass fuel and that is good for the ecosystem. Other examples of contributions of chemistry to the field of energy production are bioethanol, solar cell, insulators and gas tank.
So therefore, chemistry plays a vital role in our day to day living, it also enhances our minds for future endeavors and lastly, it can serve as an inspiration to other people in determining what field in science to prioritize because chemistry links all the sciences. Lastly, chemistry has made countless innovations for the sake of mankind.
ALWAYS REMEMBER, SAFETY IN THE LABORATORY!

All students must read and understand the information in this document with regard to laboratory rules and safety precautions prior to the first laboratory session. Your personal laboratory safety depends mostly on YOU.
Good common sense is needed for safety in a laboratory. It is expected that each student will work in a responsible manner and exercise good judgment and common sense. If at any time you are not sure how to handle a particular situation, ask your Teaching Assistant or Instructor for advice. Do not touch anything with which you are not completely familiar. It is always better to ask questions than to risk harm to yourself or damage to the equipment. Other rules in the laboratory are as follows.
You should observe proper clothing inside the laboratory. When handling dangerous substances, wear gloves, laboratory coats, shoes, and safety glasses for eye protection. It’s because we might get injured if we handle hazardous chemicals improperly. Wearing shorts and sandals are strictly prohibited inside the laboratory and if you have long hair or loose clothes, make sure it is tied back or confined.
Never eat, drink, or smoke while working in the laboratory. Smoking is prohibited because there might be a chance of accidental fire when a cigarette is close to a flammable substance you are using. Foods or drinks are also prohibited because those might be contaminated by the chemicals around you. Also, storing food in the laboratory is not allowed.
Do not play inside the laboratory because you can get clumsy and put your lab mates in serious injury.
Do not taste and smell any solvent or chemical inside the lab. There might be contents of those chemicals that are very dangerous to our body. Never allow solvents to come in contact with your skin, always use gloves and never pipette anything by mouth because it’s very dangerous.
Keep your work area clean and organized. Unneeded materials should be kept away, clearly label all chemicals that are being used and read them carefully while doing an experiment. Treat every chemical as if they were all hazardous to use them carefully and correctly.
Fix unobstructed access to all exits, fire extinguishers, electrical panels, emergency showers, and eye washes so that you can use them in case of emergency. Check your glassware for cracks and chips each time you use it. Cracks could cause the glassware to fail during use and cause serious injury to you or lab mates. Also, do not use any equipment unless you are trained and approved as a user by your supervisor.
Correct use of laboratory equipments must be observed. Every laboratory must have a first aid cabinet in case of emergency. Also remember that octopus wirings are strictly prohibited inside the lab because it can cause fire.
In disposing used substances, never return chemicals to reagent bottles. You should get the exact amount and share any excess. Dispose your waste and broken glassware in appropriate containers.
Before leaving the laboratory, clean your work area and lock the door. Never do unauthorized experiments, and If you conducting an experiment, make sure that you will not leave it unattended.
These are the laboratory rules and safety precautions that you must apply in order for you to conduct your experiment correctly and to prevent accidents from happening.
THE SCIENTIFIC NOTATION AND ITS OPERATIONS

Enough of all your complains about writing very big or very small values of numbers. There’s no time for you to make problems out of them. Don’t waste your time writing zeros which doesn’t seem to end. Why do you need to survive this situation if you can actually make it a lot more easier using scientific notation?
The scientific notation is a method of writing very big or very small values of numbers in a shorter way. In this way, numbers will be compacted. Numbers are represented by multiplying a factor to the power of 10. The power of ten is the number of 10 raised to a certain integer exponent. With this, you’ll save more time, have less work, less stressful analysis and much easier way in arriving at the accurate answer. You can also use this method in answering problems using different operations. You just need to be familiarized with the rules of this method.
In writing standard form to scientific method, you need to move the decimal point from right to left until it reaches the first non-zero digit. Then, count how many places you misplaced the decimal point. That will serve as your integer exponent.
Example:
1. Standard Form= 6 000 000 000 000 000 000 000 000
Scientific Notation= 6.0 x 10^24
2. Standard form = 0.00000000031
Scientific Notation = 3.1 x 10^-9
In writing scientific method to standard form, move the decimal point from left to right based on the integer exponent if it is positive. Move the decimal point from right to left based on the integer exponent if it is negative.
Example:
1. Scientific Notation = 7.1 x 10^6
Standard form = 7 100 000
2. Scientific Notation = 8.0 x 10^-12
Standard form = 0.000000000008
USING SCIENTIFIC NOTATION IN DIFFERENT OPERATIONS
-Addition/Subtraction
In adding or subtracting scientific notations, the integer exponent must be the same based on the bigger exponent before you can add or subtract. After they were made the same, you can then perform the operation. The integer must be obtained for the sum or difference.
Example:
Addition
9.2 x 10^8 + 6.9 x 10^7 = 9.2 x 10^8 + (6.9 x 10^7)
= 9.2 x 10^8 + 0.69 x10^8
= 9.89 x 10^8
Subtraction
9.2 x 10^8 – 6.9 x 10^7 = 9.2 x 10^8 - (6.9 x 10^7)
= 9.2 x 10^8 - 0.69 x10^8
= 8.51 x 10^8
·Multiplication
To multiply scientific notations, plainly multiply the bases. The exponents must be added. The product must be written as the product of the bases times 10 to the sum of their exponents.
Example:
(2.0 x 10^4)(1.5 x 10^3) = (2.0 x 1.5) x 10^(4+3)
= 3.0 x 10^7
-Division
In dividing scientific notations, divide the bases. The exponents must be subtracted. The quotient must be written as the quotient of bases times 10 to the difference of their exponents.
Examples:
6.0 x 10^7/3.0 x 10^4 = (6.0/3.0) x 10^(7-4)
= 2.0 x 10^3
The scientific notation is a method of writing very big or very small values of numbers in a shorter way. In this way, numbers will be compacted. Numbers are represented by multiplying a factor to the power of 10. The power of ten is the number of 10 raised to a certain integer exponent. With this, you’ll save more time, have less work, less stressful analysis and much easier way in arriving at the accurate answer. You can also use this method in answering problems using different operations. You just need to be familiarized with the rules of this method.
In writing standard form to scientific method, you need to move the decimal point from right to left until it reaches the first non-zero digit. Then, count how many places you misplaced the decimal point. That will serve as your integer exponent.
Example:
1. Standard Form= 6 000 000 000 000 000 000 000 000
Scientific Notation= 6.0 x 10^24
2. Standard form = 0.00000000031
Scientific Notation = 3.1 x 10^-9
In writing scientific method to standard form, move the decimal point from left to right based on the integer exponent if it is positive. Move the decimal point from right to left based on the integer exponent if it is negative.
Example:
1. Scientific Notation = 7.1 x 10^6
Standard form = 7 100 000
2. Scientific Notation = 8.0 x 10^-12
Standard form = 0.000000000008
USING SCIENTIFIC NOTATION IN DIFFERENT OPERATIONS
-Addition/Subtraction
In adding or subtracting scientific notations, the integer exponent must be the same based on the bigger exponent before you can add or subtract. After they were made the same, you can then perform the operation. The integer must be obtained for the sum or difference.
Example:
Addition
9.2 x 10^8 + 6.9 x 10^7 = 9.2 x 10^8 + (6.9 x 10^7)
= 9.2 x 10^8 + 0.69 x10^8
= 9.89 x 10^8
Subtraction
9.2 x 10^8 – 6.9 x 10^7 = 9.2 x 10^8 - (6.9 x 10^7)
= 9.2 x 10^8 - 0.69 x10^8
= 8.51 x 10^8
·Multiplication
To multiply scientific notations, plainly multiply the bases. The exponents must be added. The product must be written as the product of the bases times 10 to the sum of their exponents.
Example:
(2.0 x 10^4)(1.5 x 10^3) = (2.0 x 1.5) x 10^(4+3)
= 3.0 x 10^7
-Division
In dividing scientific notations, divide the bases. The exponents must be subtracted. The quotient must be written as the quotient of bases times 10 to the difference of their exponents.
Examples:
6.0 x 10^7/3.0 x 10^4 = (6.0/3.0) x 10^(7-4)
= 2.0 x 10^3
DISCOVER THE ELEMENTS AND KNOW THE COMPOUNDS

You’ve probably heard about Elements and Compounds especially when discussing about different substances on Chemistry. But did you ever wonder what is the difference between these both terms? Let's find -out!
Let us first discover the elements…
Elements are commonly found in a table in chemistry. There is one table that contains 120 elements so far and that is the Periodic Table of Elements, it is considered to be the heart and soul of Chemistry. To be familiar, elements are substances that cannot be separated into simpler substance because it is already in its simplest form. Also, it contains only one kind of atom. They can also exist as either atom like argon or molecules like nitrogen. Copper for example will remain as copper for it is a known element. Other substances are also made up of several elements. Chemists use symbols to represent elements. A symbol is a letter or picture used to represent something. Chemists use one or two letters to represent elements. The symbol for aluminum is Al. The symbol for oxygen is O and the symbol of Livermorium is Lv.
Elements can be classified as metals, non-metals and metalloids. Metals are elements that easily conduct electricity and as of now, there are 91 out of 120 elements in the Periodic table. Metals are malleable, fusable and ductile. Examples of metals are the well-known gold, silver, aluminum and iron.
Let us now discover non-metals. Non-metals have no capability to conduct electricity and don’t conduct heat very well. They cannot be pounded into thin sheets or wires. For short, they are neither malleable nor ductile. They have no luster and they do not possess the ability to reflect light. Known examples of non-metals are carbon and gases like carbon, oxygen, nitrogen etc.
Last are the metalloids. Metalloids are the elements found along the stair-step line that distinguishes metals from non-metals. This line is drawn from between Boron and Aluminum to the border between Polonium and Astatine. The only exception to this is Aluminum, which is classified under "Other Metals". Metalloids have properties of both metals and non-metals. Some of the metalloids, such as silicon and germanium, are semi-conductors. This means that they can carry an electrical charge under special conditions. This property makes metalloids useful in computers and calculators
Now, let us step forward to know what are compounds…
A compound is a substance formed when two or more elements are chemically joined. Water (H2O), two hydrogen and 1 oxygen atom, salt (NaCl) and sugar (C12H22O11) are examples of compounds. Compounds can be separated into their original composition by chemical means but never by physical means. Chemical bonds link elements together to form more complex molecules. A compound consists of two or more types of elements held together by covalent or ionic bonds.
When the elements are joined, the atoms lose their individual properties and have different properties from the elements they are composed of. A chemical formula is used a quick way to show the composition of compounds. Letters, numbers, and symbols are used to represent elements and the number of elements in each compound.
There are two classification of compounds based on composition: Organic compound which contain carbon like sugar and inorganic compound or substances with the absence of carbon like salt.
Let us first discover the elements…
Elements are commonly found in a table in chemistry. There is one table that contains 120 elements so far and that is the Periodic Table of Elements, it is considered to be the heart and soul of Chemistry. To be familiar, elements are substances that cannot be separated into simpler substance because it is already in its simplest form. Also, it contains only one kind of atom. They can also exist as either atom like argon or molecules like nitrogen. Copper for example will remain as copper for it is a known element. Other substances are also made up of several elements. Chemists use symbols to represent elements. A symbol is a letter or picture used to represent something. Chemists use one or two letters to represent elements. The symbol for aluminum is Al. The symbol for oxygen is O and the symbol of Livermorium is Lv.
Elements can be classified as metals, non-metals and metalloids. Metals are elements that easily conduct electricity and as of now, there are 91 out of 120 elements in the Periodic table. Metals are malleable, fusable and ductile. Examples of metals are the well-known gold, silver, aluminum and iron.
Let us now discover non-metals. Non-metals have no capability to conduct electricity and don’t conduct heat very well. They cannot be pounded into thin sheets or wires. For short, they are neither malleable nor ductile. They have no luster and they do not possess the ability to reflect light. Known examples of non-metals are carbon and gases like carbon, oxygen, nitrogen etc.
Last are the metalloids. Metalloids are the elements found along the stair-step line that distinguishes metals from non-metals. This line is drawn from between Boron and Aluminum to the border between Polonium and Astatine. The only exception to this is Aluminum, which is classified under "Other Metals". Metalloids have properties of both metals and non-metals. Some of the metalloids, such as silicon and germanium, are semi-conductors. This means that they can carry an electrical charge under special conditions. This property makes metalloids useful in computers and calculators
Now, let us step forward to know what are compounds…
A compound is a substance formed when two or more elements are chemically joined. Water (H2O), two hydrogen and 1 oxygen atom, salt (NaCl) and sugar (C12H22O11) are examples of compounds. Compounds can be separated into their original composition by chemical means but never by physical means. Chemical bonds link elements together to form more complex molecules. A compound consists of two or more types of elements held together by covalent or ionic bonds.
When the elements are joined, the atoms lose their individual properties and have different properties from the elements they are composed of. A chemical formula is used a quick way to show the composition of compounds. Letters, numbers, and symbols are used to represent elements and the number of elements in each compound.
There are two classification of compounds based on composition: Organic compound which contain carbon like sugar and inorganic compound or substances with the absence of carbon like salt.
mixtures and their characteristics
All of us are familiar about the term mixture. It is mostly used in cooking. But now, it is the mixture in Chemistry that you’re going to learn. Mixture is absolutely everywhere you look. The ocean, soil, alcohol and catsup are all examples of mixtures. But how will you know if a certain substance is a mixture? That is what we are going to talk about.
In chemistry, a mixture is made up of two or more different substances which are physically mixed together but not chemically combined. It is the combination of two or more substances wherein the identities of the combined substances were maintained. But even though there are no chemical changes to its constituents, the physical properties of a mixture, such as its melting point, may differ from those of the components.
But how can we know if a certain substance is a mixture?
Most of us are confused in the difference of mixture and compound. But the answer is just right in front of you and it is only simple. We can consider a substance as mixture if the substances are mixed together but not chemically combined. And compounds are substances that contain two or more elements that were chemically combined. For example, a cake dough and a baked cake. Of course, a cake dough is a mixture because all of the ingredients were only combined physically or mixed together to form it. There is no chemical changes happened so it is considered as a mixture. But when you bake the cake dough, of course, you will use heat to cook it which means there will be a chemical change that will happen because all of the ingredients will be mixed chemically.
Now that you know the difference of mixture and compound, it’s time for you to learn the two types of mixtures: homogeneous and heterogeneous.
Homogeneous mixtures are substances that have uniform composition and properties. It is an object that has the same composure throughout. It means that when dividing the volume in half, the same amount of material is suspended in both halves of the substance. This type of mixture is very stable, its particles do not settle, or separate. Homogeneous mixtures have only one phase, meaning, the physical properties of the whole substance itself is essentially uniform.
How can we know if a certain substance is a homogeneous mixture?
A substance is a homogeneous mixture if the different substances that were physically combined have one phase or state and when the two ingredients are not separately identifiable when you look at it. For example, salt water. Salt water is considered as a homogeneous mixture because it is made up of different substances but the other substance became invisible to form one phase but the substances retained its own physical and chemical identity.
Heterogeneous mixture is a type of mixture in which the components can be seen, as there are two or more phases present. It is a mixture of dissimilar items. One substance can be hydrophilic and the other one can be hydrophobic, in which they can’t be mixed together so they will remain physically separated. They always have more than one phase.
How can we know if a certain substance is a heterogeneous mixture?
We can determine if a substance is a heterogeneous if there are more than one phase and when they are still physically separated when mixed together. For example, salad. Salad is considered as a heterogeneous mixture because you can clearly see that the ingredients mixed together were still visible and you can distinguish what ingredient is it. Another example, a glass of iced tea is a heterogeneous mixture. The ice cubes have different composition and properties than the tea. You can say that the ice cubes are in solid phase, and the tea is in liquid phase. Liquids and solids cannot be mixed together unless one of them changes its phase. From that, we can say that ice tea is a heterogeneous.
THE WORLD OF SOLUTIONS, SUSPENSIONS & COLLOIDS

A Solution is a Homogenous Mixture that is made up of only one phase. It is a groups of molecules that are mixed up in a completely even distribution. Solutions can be solids dissolved in liquids. When you work with chemistry or even cook in your kitchen, you will usually be dissolving solids into liquids. Solutions could also be gases dissolved in liquids (such as carbonated water). There can also be gases in other gases, and liquids in liquids. If you mix things up and they stay at an even distribution, it is a solution. You probably won't find people making solid-solid solutions. They usually start off as solid/gas/liquid-liquid solutions and then harden at room temperature. Alloys with all types of metals are good examples of a solid solution at room temperature.
EXAMPLES OF SOLUTIONS:
Air
Carbon Dioxide (CO2) in Soda
Hydrogen (H2) in Palladium (Pd) Metal
Gasoline
Dental Fillings
Metal Alloys Such as Sterling Silver
A simple solution is basically two substances that are going to be combined. One of them is called the solute. A solute is the substance to be dissolved (sugar). The other is a solvent. The solvent is the one doing the dissolving (water). As a rule of thumb, there is usually more solvent than solute. Be patient with the next sentence as we put it all together. The amount of solute that can be dissolved by the solvent is defined as solubility.
A Suspension is a Heterogeneous mixture, meaning that it has clearly separated parts and is not chemically combined. It is a mixture between two substances, one of which is finely divided and dispersed in the other. Common suspensions include sand in water, dust in air, and droplets of oil in air. Particles in a suspension are larger than those in a solutions; they are visible under a microscope and can often be seen with the naked eye. Particles in a suspension will settle out if the suspension is allowed to stand undisturbed. Many particles of a suspension can be separated through a filter. An example of a simple suspension would be flour in water, or sand in water
Examples of Suspensions
1. Sometimes, in the right light, you will be able to see particles of dust floating in a room. Eventually the dust will settle on the floor and on furniture, and the room will need to be cleaned. Dust in air is a suspension.
2. If you go to a beach and mix sand and water in a bucket you will form a suspension. Given time, the sand will settle on the bottom of the bucket to leave clear water.
A colloid is a type of mixture intermediate between a homogeneous mixture and a heterogeneous mixture with properties also intermediate between the two.The particles in a colloid can be solid, liquid or bubbles of gas. The medium that they are suspended in can be a solid, liquid or gas (although gas colloids cannot be suspended in gas).The particles are approximately 10 to 10,000 angstroms in size and generally cannot be filtered, or settled out in an easy manner. Colloids may be colored or translucent because of the Tyndall effect, which is the scattering of light by particles in the colloid. Colloid particles may be seen in a beam of light such as dust in air in a "shaft" of sunlight.
An easy way of determining whether a mixture is colloidal or not is through use of the Tyndall Effect. When light is shined through a true solution, the light passes cleanly through the solution, however when light is passed through a colloidal solution, the substance in the dispersed phases scatters the light in all directions, making it readily seen. An example of this is shining a flashlight into fog. The beam of light can be easily seen because the fog is a colloid. Light being shined through water and milk. The light is not reflected when passing through the water because it is not a colloid. It is however reflected in all directions when it passes through the milk, which is colloidal.
DETERMINING AND KNOWING ACIDS, BASES AND NEUTRAL SUBSTANCES

There are so many kinds of substance and chemicals you can see in your chemistry laboratory and even in your everyday household; different in color, odor, taste and of course, composition. Some may be good for your health, some may cause irritation, or even be toxic. They can be ACID, BASE or NEUTRAL. What is an acid, base and neutral? How can you distinguish them? That’s what will you found out after reading this.
Let’s start with ACID. You may heard this a lot of times before, but did you know the real meaning of this term? Acid came from the Latin word acidus, which means sour. All Acids are generally sour in taste. It has a pH level of less than 7. Acids may be categorized by weak acid with 1.6-6.9 pH level and strong acid with 0-1.5 pH. It conducts electricity and reacts with metals to form H2 gas. Acids can also be characterized by having the presence of hydrogen ions.
Here are some examples of acids with their chemical formula:
The “opposite” of an acid is BASE. Bases taste bitter and feel slippery. It has a negatively charged molecular fragment consisting of an oxygen atom and a hydrogen atom (OH-). This fragment is called a hydroxide ion. Bases are also categorized by weak and strong bases. The more accessible a bases hydroxide ion is, the stronger it is. Combining a strong base and strong acid will result in vigorous reactions.
The combination of acid (H+) and base (OH-) will produce a NEUTRAL SUBSTANCE like water, which has no change. This process is called neutralization.
Since water contains both H+ and OH- it behaves as both an acid or as a base.
Acid-Base Indicators
*Red and Blue Litmus Paper
Litmus is a water-soluble mixture of different dyes extracted from lichens, especially Roccella tinctoria. It is often absorbed onto filter paper to produce one of the oldest forms of pH indicator, used to test materials for acidity.
When Red litmus paper remains the same and blue litmus paper turns red, this means that the substance dropped to the litmus paper is an acid. And when Blue litmus paper remains the same and Red litmus paper turns blue, the substance is a base.
*Bromothymol Blue
Bromothymol blue is a pH indicator for weak acids and bases. It is mostly used in applications that require measuring substances that would have a relatively neutral pH, such as managing the pH of pools and fish tanks.
Bromothymol blue turns yellow when a base is dropped and blue for acid . It is green at pH 7.0 which is the neutralization pH of a weak acid/weak base titration.
*Universal Indicator
A Universal indicator is a pH indicator composed of a solution of several compounds that exhibits several smooth colour changes over a pH value range from 1-14 to indicate the acidity or alkalinity.
*Phenolphthalein
Phenolphthalein is a chemical compound with the formula C20H14O4 and is often written as "HIn" or "phph" in shorthand notation. Often used in titrations, it turns colorless in acidic solutions and pink in basic solutions. If the concentration of indicator is particularly strong, it can appear purple. In strongly basic solutions, phenolphthalein's pink color undergoes a rather slow fading reaction and becomes colorless again.
*Methyl Orange
Methyl orange is a pH indicator frequently used in titrations. It is often used in titrations because of its clear and distinct colour change. Because it changes colour at the pH of a mid-strength acid, it is usually used in titrations for acids.
Methyl orange turns to orange when an acid is dropped and remains the same or yellow when reacts with base.
*Methyl Red
Methyl red, also called C.I. Acid Red 2, is an indicator dye that turns red in acidic solutions. It is an azo dye, and is a dark red crystalline powder.
This indicator turns red when reacts with acid, and when turns yellow it is a base.
Let’s start with ACID. You may heard this a lot of times before, but did you know the real meaning of this term? Acid came from the Latin word acidus, which means sour. All Acids are generally sour in taste. It has a pH level of less than 7. Acids may be categorized by weak acid with 1.6-6.9 pH level and strong acid with 0-1.5 pH. It conducts electricity and reacts with metals to form H2 gas. Acids can also be characterized by having the presence of hydrogen ions.
Here are some examples of acids with their chemical formula:
The “opposite” of an acid is BASE. Bases taste bitter and feel slippery. It has a negatively charged molecular fragment consisting of an oxygen atom and a hydrogen atom (OH-). This fragment is called a hydroxide ion. Bases are also categorized by weak and strong bases. The more accessible a bases hydroxide ion is, the stronger it is. Combining a strong base and strong acid will result in vigorous reactions.
The combination of acid (H+) and base (OH-) will produce a NEUTRAL SUBSTANCE like water, which has no change. This process is called neutralization.
Since water contains both H+ and OH- it behaves as both an acid or as a base.
Acid-Base Indicators
*Red and Blue Litmus Paper
Litmus is a water-soluble mixture of different dyes extracted from lichens, especially Roccella tinctoria. It is often absorbed onto filter paper to produce one of the oldest forms of pH indicator, used to test materials for acidity.
When Red litmus paper remains the same and blue litmus paper turns red, this means that the substance dropped to the litmus paper is an acid. And when Blue litmus paper remains the same and Red litmus paper turns blue, the substance is a base.
*Bromothymol Blue
Bromothymol blue is a pH indicator for weak acids and bases. It is mostly used in applications that require measuring substances that would have a relatively neutral pH, such as managing the pH of pools and fish tanks.
Bromothymol blue turns yellow when a base is dropped and blue for acid . It is green at pH 7.0 which is the neutralization pH of a weak acid/weak base titration.
*Universal Indicator
A Universal indicator is a pH indicator composed of a solution of several compounds that exhibits several smooth colour changes over a pH value range from 1-14 to indicate the acidity or alkalinity.
*Phenolphthalein
Phenolphthalein is a chemical compound with the formula C20H14O4 and is often written as "HIn" or "phph" in shorthand notation. Often used in titrations, it turns colorless in acidic solutions and pink in basic solutions. If the concentration of indicator is particularly strong, it can appear purple. In strongly basic solutions, phenolphthalein's pink color undergoes a rather slow fading reaction and becomes colorless again.
*Methyl Orange
Methyl orange is a pH indicator frequently used in titrations. It is often used in titrations because of its clear and distinct colour change. Because it changes colour at the pH of a mid-strength acid, it is usually used in titrations for acids.
Methyl orange turns to orange when an acid is dropped and remains the same or yellow when reacts with base.
*Methyl Red
Methyl red, also called C.I. Acid Red 2, is an indicator dye that turns red in acidic solutions. It is an azo dye, and is a dark red crystalline powder.
This indicator turns red when reacts with acid, and when turns yellow it is a base.
REFERENCES OF INFORMATION AND PICTURES
- WE would like to acknowledge Google.com for all the pictures and valid resource information for the completion of the articles.
- Especially we would also like to thank www.chemistry.about.com and www.wikipedia .com.
- For the pictures of our members we would like to the cite the source which is www.facebook.com
- And for the "Cool Trivia" section we would like to thank www.sciensational.com/chemistry.html and chemistry.about.com › Education › Chemistry › Chemistry Facts